Jadeite Molecular Structure
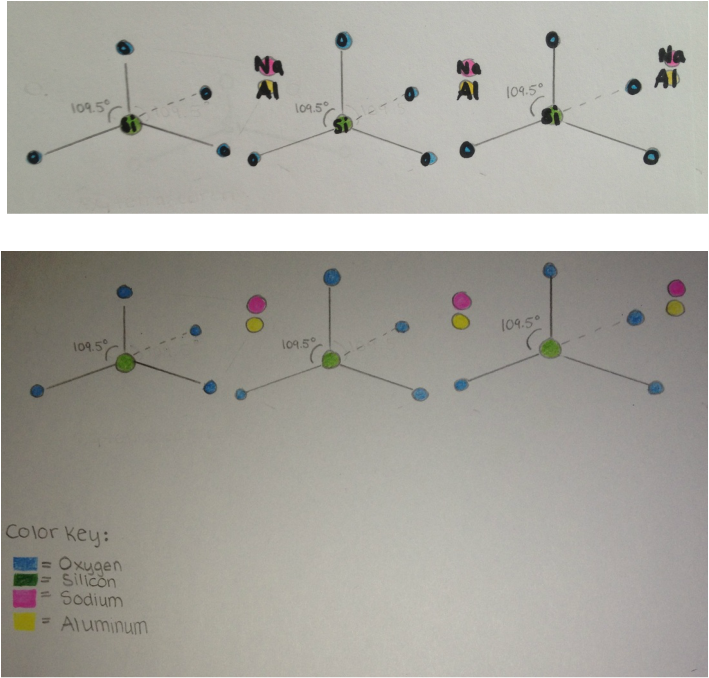
As shown in the drawings below, jadeite's structure is repeating, made up of SiO4 molecules that are attracted to Na and Al atoms. Within the molecule, one silicone atom bonds to four oxygen atoms, forming an AX4 tetrahedron. The bond angles of the tetrahedron are 109.5º. The molecule is polar because its structure is asymmetrical, and the molecule does not exhibit an equal distribution of charges.
